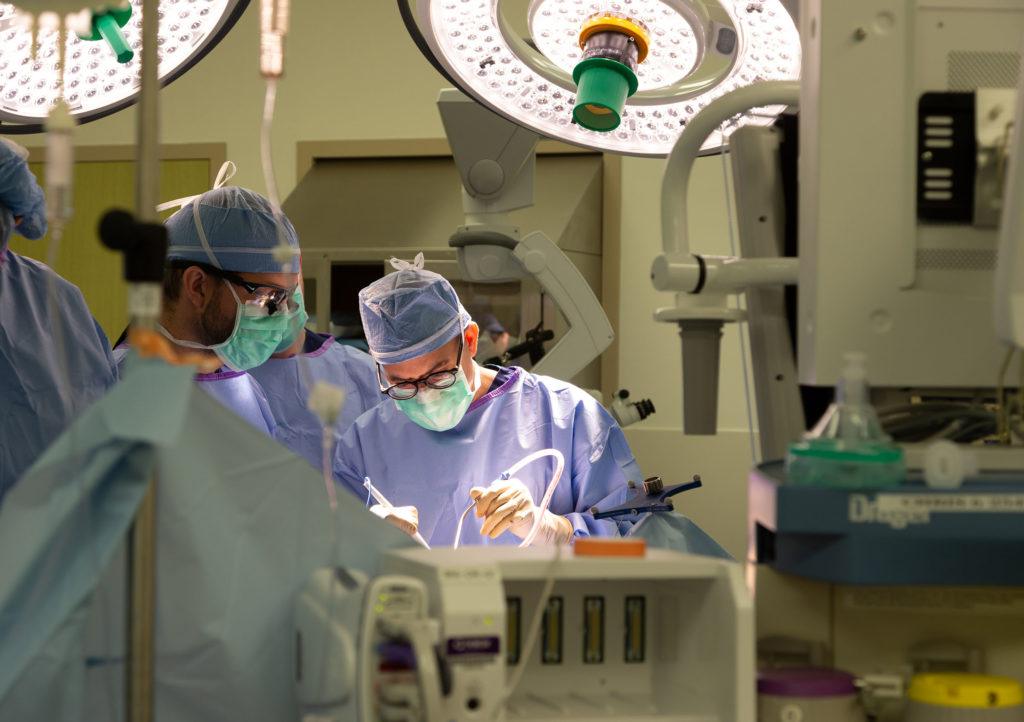
High-Grade Glioma and Glioblastoma Clinical Trials
What is a glioma Brain tumor?
Gliomas are primary brain tumors, which means they are derived from the brain tissue itself. Common subtypes include astrocytoma, oligodendroglioma, glioblastoma, and gliosarcoma.
All gliomas are graded based upon World Health Organization (WHO) criteria:
Grades II, III, and IV gliomas are most common in adults. Grades I, II, and III gliomas are less aggressive and often seen in younger populations, yet can still recur and defy conventional therapies. All gliomas cause symptoms through a combination of mass effect on the surrounding brain and direct infiltration of the brain tissue.
What is a high-grade glioma brain tumor?
A high-grade glioma or Grade IV gliomas (known as glioblastomas or gliosarcomas) are the most common primary brain cancer in adults. Glioblastoma is a rapidly growing tumor that can occur at any age, but its incidence increases with advanced age.
Gliomas are a primary focus of the Ivy Brain Tumor Center and a large proportion of our clinical trials portfolio focuses on high-grade gliomas.
What is a recurrent glioblastoma brain tumor?
A glioblastoma that appears to have returned after a patient has undergone initial treatment is called a recurrence.
Even after maximal surgical resection, microscopic glioblastoma cells often remain and can grow back over time. To slow their growth, patients are treated with the current standard of care which consists of temozolomide chemotherapy and radiation therapy. However, standard treatment is not curative and most patients experience tumor progression.To monitor for tumor regrowth, patients are scheduled for follow-up visits and routine MRI scans. Regardless of whether you are newly diagnosed or have been informed by a doctor that your tumor has recurred (come back), you may be eligible for an Ivy Phase 0 clinical trial.
World Health Organization (WHO) Criteria
Slow-growing tumor cells; almost normal appearance; least aggressive
Relatively slower-growing cells; slightly abnormal appearance; often invade nearby tissue; may recur as a higher-grade tumor
Faster-dividing cells; abnormal appearance; infiltrates normal tissue and often recur as higher-grade tumor
Rapidly-dividing and abnormal cells; contains necrotic areas of nonviable tissue
Glioma and Glioblastoma Symptoms
Glioblastoma symptoms vary widely depending on the type, location, size and growth rate of the brain tumor. There are no specific symptoms that only occur due to a glioma tumor.
General symptoms of brain tumors include:
- New onset or change in pattern of headaches
- Headaches that gradually become more frequent and more severe
- New onset of seizures
- Gradual loss of sensation or movement in an arm or a leg
- Difficulty with balance
- Difficulty speaking
- Personality or behavior changes
- Confusion
- Unexplained nausea or vomiting
- Blurred vision, double vision, or loss of peripheral vision
- Hearing problems
Contact your physician if you are experiencing any of these symptoms.
RECURRENT GLIOblastoma phase 0 clinical trials
If you have a glioma and undergone MR imaging that shows evidence of tumor recurrence, you may be a candidate for one of our Phase 0 clinical trials. Our Phase 0 studies provide you with information you can use right now to make informed treatment decisions. This knowledge allows you to have a voice in your care and places some control back into your hands.
Stay Informed
Sign up to be alerted when new trials open.
Medically Reviewed by: Dr. Nader Sanai
Am I Eligible?
View a list of our open trials and receive a free trial screening to determine your eligibility.